How to clean dirty coins
by Ailsa Harvey · 31/03/2021
Remove the oxide layer from the outside of dirty coins using fruit juice!

1. Find some coins
First, you’ll need to find some coins to clean. Newer coins will already be very clean, so you’re looking for older ones if possible. Coppers like pennies or 2p pieces are best for this, but you can use silver coins if necessary. Try to find coins that are really dirty or even a little bit tarnished so that you will really be able to see the difference cleaning can make.

2. Clip them on
We’re only going to submerge half the coin- the other half will not touch the liquid. This means the weak acids in the cleaning liquid will not affect the other half, so you’ll be able to see the difference. You need a strong clip to secure the coins above the glass, so use a bulldog clip to hold the coin and push a pencil through the top to balance your clip across a glass.

3. Prepare your cleaner
You might be surprised to hear that the cleaning liquid we’re going to use is actually apple juice. The juice contains citric acid, a weak acid that can help to clean coins. The coin is dull because the copper in the metal has reacted with oxygen in the air to form copper oxide. This oxide makes the coin look dirty, but the citric acid in the juice will get rid of it and restore the coin’s shine.
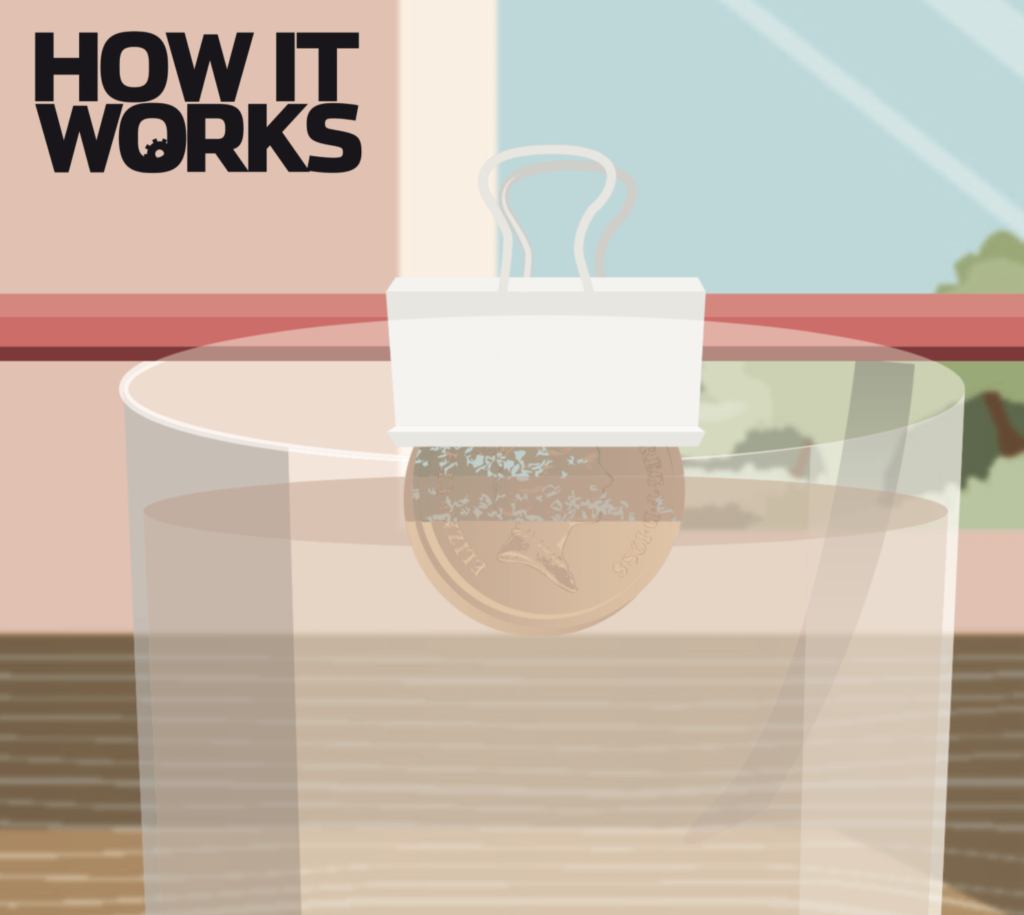
4. The waiting game
You need to pour the fruit juice into the glass so that it only covers the bottom half of the coin. Don’t worry if you happen to put too much in- just pour a little bit out of the glass until the level is right. You now need to leave the coin in the fruit juice for at least a few hours- if possible, leave it overnight for the best results. Then, remove the coin to see how effective the cleaner is!

5. Other cleaners
You can use other liquids to clean your coins, too. Different fruit juices contain different levels of acid, so experiment with a variety of options to find which is the most effective. You can also try liquids like ketchup, which contains vinegar, or just vinegar on its own. Vinegar contains acetic acid, which is quite strong, so it should do a great job of cleaning your coins.
Summary
When metals react with oxygen in water or air, they form oxides. These oxides are often referred to as ‘rust’. Weak acids can dissolve metal oxides, allowing you to restore the shine of coins. However, doing so will remove a very thin outer layer of the coin, so if you did it too much you wouldn’t have a coin left!
For more science and technology articles, pick up the latest copy of How It Works from all good retailers or from our website now. If you have a tablet or smartphone, you can also download the digital version onto your iOS or Android device. To make sure you never miss an issue of How It Works magazine, subscribe today!









